Podcast: Play in new window | Download
“This learning material is sourced from Emergency Medicine Cases and has been published here with permission as per Creative Commons copyright.”
Topics in this EM Quick Hits podcast
Kate Lazier on a case of fever without a source (1:13)
Andrew Petrosoniak on chest tubes in trauma part 2 (15:59)
Adrianna Roweon the challenges of recognition and management of medetomidine withdrawal (28:49)
Justin Morgensternon why the PECARN c-spine rule may be dangerous (36:19)
Zach Cantor on paramedic handover done right (48:45)
Sara Gray & Katie Lin on coaching the em mind part 1: physiology – sleep, nutrition and exercise (59:16)
Podcast production, editing and sound design by Anton Helman
Podcast content, written summary & blog post by Anton Helman, March, 2026
Cite this podcast as: Helman, A. Lin, K. Lazier, K. Petrosoniak, A. Morgenstern J. Cantor, Z. Gray, S. EM Quick Hits 71 – EMC²: Fever Without a Source, Coaching the EM Mind Part 1, Traumatic Pneumothorax Part 2, PECARN C-spine Rule, Medetomidine Withdrawal, EMS Handover. March, 2026. https://emergencymedicinecases.com/em-quick-hits-march-2026/. Accessed March 11, 2026.
EM Cases Cases (EMC²): Fever Without an Obvious Source and Fever of Unknown Origin – Don’t Miss the Treatable Diagnoses!
- When evaluating fever without a clear source, prioritize treatable, life/limb/vision-threatening diagnoses especially ones that are easy to miss.
- Address epidemiologic risk factors including travel, sexual exposure, immunocompromised states, outdoor exposures, and substance use, as these may significantly narrow the diagnostic differential.
High-risk infectious diagnoses to consider
- Endocarditis
- Meningitis
- Spinal epidural abscess
- Occult abscesses
Inflammatory diagnoses
-
Giant cell arteritis and other vasculitites
Malignancy
-
Leukemia / blast crisis
Toxicologic / endocrine diagnoses
- Serotonin syndrome
- Neuroleptic malignant syndrome
- Alcohol withdrawal
- Stimulant intoxication
- Thyroid storm
- Adrenal insufficiency
- Salicylate toxicity
West Nile Virus pearls
- Endemic in Canada for ~20 years
- Most infections asymptomatic
- Symptomatic cases → fever + malaise
- Severe cases → encephalitis, flaccid paralysis, Parkinsonian features
- Treatment is supportive
Fever of Unknown Origin (as apposed to Fever Without a Source)
- Defined as ≥38.3°C for ≥3 weeks
- Up to 50% never receive a diagnosis, with most of these patients ultimately doing well
- Ensure appropriate follow-up if discharged
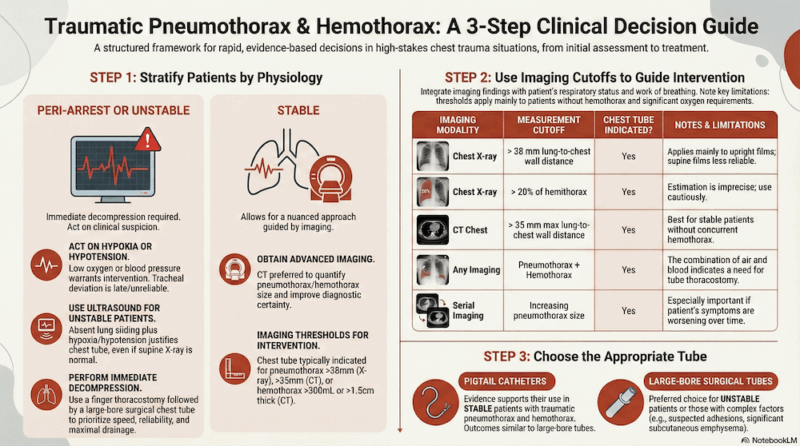
Traumatic Pneumothorax Part 2: Chest Tube Decision Pearls
If you haven’t already, listen to Part 1 first where Dr. Petrosoniak talks about the following…
Needle Decompression in the ED for traumatic pneumothorax
- Needle decompression is increasingly recognized as unreliable in the emergency department because chest wall thickness often exceeds the length of standard angiocatheters, resulting in frequent failure to enter the pleural space and delaying definitive treatment.
- Even when performed in the lateral chest (4th–5th intercostal space), success remains unpredictable; therefore critically ill trauma patients should not rely on this temporizing maneuver when definitive decompression is required.
- In unstable patients with suspected tension physiology, the preferred approach is immediate finger thoracostomy followed by insertion of a large-bore surgical chest tube, while needle decompression may still have a limited role in prehospital or resource-limited settings where tube thoracostomy cannot be performed.
Physiology-First Approach to Traumatic Pneumothorax
- Management should begin with physiologic risk stratification, categorizing patients as peri-arrest, unstable, or stable, as physiology determines urgency of intervention, imaging strategy, and chest tube selection as outlined in Part 1.
- Peri-arrest or unstable patients, such as those with hypoxia or hypotension after chest trauma, require immediate decompression with finger thoracostomy and placement of a large-bore chest tube (typically 24–28 Fr), recognizing that classic signs of tension pneumothorax are often late and that bedside ultrasound demonstrating absent lung sliding in an unstable patient should prompt rapid intervention.
- Stable patients may be managed more selectively with imaging and clinical integration; pigtail catheters are increasingly used for isolated traumatic pneumothorax in stable patients, although large-bore tubes remain appropriate in more complex trauma or when hemothorax is suspected.
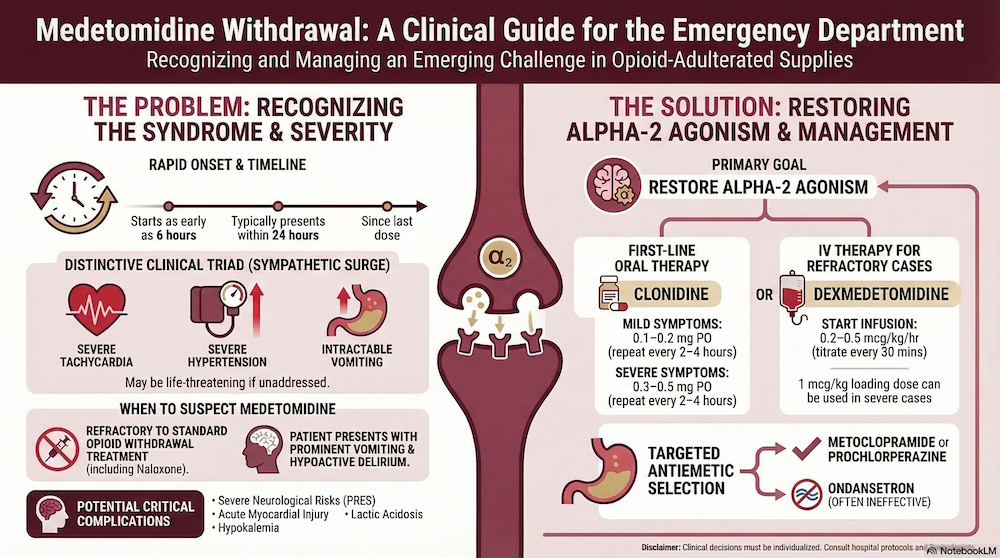
Medetomidine Withdrawal: A New and Challenging Withdrawal Syndrome to Recognize and Manage
Medetomidine, a veterinary sedative and potent non-selective alpha-2 adrenergic agonist pharmacologically similar to clonidine and dexmedetomidine, has increasingly been identified as an adulterant in the unregulated opioid supply and is now emerging as an important cause of withdrawal syndromes presenting to emergency departments.
Acute toxicity from alpha-2 agonists typically produces sedation and sinus bradycardia, and although naloxone may reverse concomitant opioid-induced respiratory depression, it does not reverse sedation related to alpha-2 agonists or benzodiazepines that may also be present in contaminated drug supplies.
The more challenging clinical presentation is medetomidine withdrawal…
Medetomidine Withdrawal Syndrome (can begin within 6 hours)
Key features:
- Severe tachycardia
- Severe hypertension
- Mydriasis
- Hypoactive delirium
- Intractable vomiting
Complications reported:
- Posterior reversible encephalopathy syndrome (PRES)
- Myocardial injury
- Hypokalemia
- Lactic acidosis
When to Suspect Medetomidine Withdrawal
Patients using unregulated drugs with:
- Severe HTN/tachycardia refractory to opioid withdrawal treatment
- Prominent vomiting
- Hypoactive delirium
- Symptoms beginning within 24 hrs of last use
Treatment of Medetomidine Withdrawal
The goal of medetomidine withdrawal treatment is to restore alpha-2 agonism
First line (if oral tolerated):
Clonidine
- Mild symptoms: 0.1–0.2 mg PO
- Severe symptoms: 0.3–0.5 mg PO
- Repeat every 2–4 hrs
If refractory or cannot take PO:
Dexmedetomidine infusion
- Start 0.2–0.5 mcg/kg/hr
- Titrate every 30 min
- Loading dose 1 mcg/kg may be used in severe cases
Antiemetic choices in management of medetomidine withdrawal
Use antidopaminergic agents
- Metoclopramide
- Prochlorperazine
Note that ondansetron is often ineffective.
Pediatric PECARN C-Spine Rule: Important Study, Limited Clinical Application
A recent multicenter study derived and validated a pediatric c-spine decision rule for children with blunt trauma presenting to trauma centers, identifying several high-risk clinical features associated with a substantially increased likelihood of cervical spine injury.
High-risk findings include:
- severely depressed consciousness,
- unresponsiveness,
- need for airway, breathing, or circulatory intervention, and
- focal neurologic deficit
Additional intermediate-risk features include:
- self-reported neck pain,
- altered mental status,
- substantial head or torso trauma, and
- neck tenderness on examination.
Despite the methodological riggor of the study, the resulting rule demonstrates only moderate sensitivity, raising concern that a clinically meaningful proportion of injuries could be missed, while the positive predictive value remains extremely low given the rarity of cervical spine injury even in high-risk trauma populations.
Importantly, the rule was derived in pediatric trauma center cohorts with far higher baseline imaging rates than most emergency departments, and has not yet undergone adequate external validation, comparison with clinician judgment, or implementation studies assessing its real-world impact on imaging practices.
In the opinion of our guest expert, routine application of the rule outside highly selected trauma settings may paradoxically increase cervical spine imaging, potentially exposing large numbers of children to unnecessary radiation without improving diagnostic accuracy.
IMIST-AMBO – Structured EMS Handover: A Critical Patient Safety Intervention
Communication during prehospital-to-emergency department handover represents a critical but frequently under-recognized patient safety intervention, with observational studies demonstrating substantial information loss during transitions of care.
Research suggests that a significant proportion of key clinical information is either omitted or not retained during handover, with frequent interruptions, parallel conversations, and premature patient transfer contributing to incomplete communication and potential downstream impacts on patient care.
Evidence consistently demonstrates that handovers are most effective when they are brief, structured, and delivered before patient transfer, allowing receiving teams to focus attention on the information being communicated.
The IMIST-AMBO framework provides a standardized structure for handover:
IMIST
- I – Identification
- M – Mechanism/medical complaint
- I – Injuries / information
- S – Signs (vitals, GCS)
- T – Treatments and trends
AMBO
- A – Allergies
- M – Medications
- B – Background
- O – Other relevant information
EMS Handover Pearls
- Begin before moving the patient off of the EMS stretcher and onto the ED stretcher
- Ensure hands off, eyes on to whomever is speaking
- Identify who is receiving report and silence the room otherwise
- Use a structured format every time
- Keep it short (~60 seconds)
Bottom Line -> Better EMS handover → fewer interruptions, clearer communication, improved patient care.
Coaching the EM Mind Part1: Physiology – Sleep, Nutrition & Exercise
Emergency medicine places clinicians in a uniquely demanding cognitive and physiologic environment characterized by shift work, sleep disruption, high cognitive load, and repeated exposure to emotionally stressful situations. Over time, these stressors can degrade both clinical performance and personal wellbeing, making deliberate attention to physiologic needs an essential component of professional sustainability.
Emergency medicine practice exposes clinicians to sustained physiologic and psychological stressors, particularly related to shift work, which is associated with increased risk of metabolic syndrome, cardiovascular disease, malignancy, and mental health disorders.
In addition to long-term health risks, sleep disruption from rotating shift schedules impairs cognitive performance, concentration, reaction time, and clinical decision-making, with fatigue-related motor vehicle collisions representing an additional hazard during post-shift commutes.
Strategies to mitigate these effects include:
Recognizing Early Warning Signs
Early indicators of declining resilience often emerge in four physiologic domains: sleep, physical conditioning, nutrition, and stress regulation. Warning signs may include falling asleep during commutes or routine activities, progressive deterioration in physical fitness or health metrics, irregular or inadequate nutrition during shifts, and increasing feelings of overwhelm, irritability, or dread before work. Importantly, these changes often develop gradually and may be more readily recognized by colleagues or family members before clinicians recognize them themselves.
Scheduling Strategies to Mitigate Circadian Disruption
Several evidence-informed scheduling principles may help mitigate these effects. Whenever feasible, schedules should incorporate flexibility that allows clinicians to align shifts with their chronotype, recognizing that individuals naturally differ in their sleep–wake preferences.
- Forward-rotating schedules (day to evening to night) are generally better tolerated than backward rotation, as they align more closely with natural circadian adjustment.
- Shorter shifts—often seven to eight hours—may reduce cognitive fatigue and error rates, while adequate recovery time between shifts, ideally more than eleven hours, allows for meaningful physiologic restoration.
- Transparent and equitable scheduling practices further support psychological wellbeing by reinforcing fairness and professional agency.
Optimizing Sleep for Shift Workers
Because sleep disruption is unavoidable in emergency medicine, clinicians benefit from intentionally optimizing sleep environments and routines. Practical measures include maintaining a dark, cool, and quiet sleeping environment, using blackout blinds, eye masks, earplugs, and white-noise devices when necessary. Strategic pre-shift napping before night shifts, minimizing caffeine intake in the hours preceding sleep, and limiting exposure to bright light before planned sleep periods may also improve sleep quality. Developing consistent routines around sleep—even when schedules vary—can significantly improve restorative sleep over time.
Episode 207 Deep dive into sleep strategies for shift workers
Exercise and Cognitive Performance
Regular physical activity plays a critical role in maintaining both physical and cognitive resilience. Exercise has been shown to improve attention, memory, mood regulation, and stress tolerance, while also reducing long-term risks of cardiovascular disease and dementia. Even modest and consistent exercise routines—such as brief daily workouts or incorporating activity into existing routines—can yield meaningful physiologic and psychological benefits. Importantly, sustainable habits often begin with small, achievable goals rather than ambitious but unsustainable lifestyle changes.
Nutrition and Cognitive Function
Nutrition is an often overlooked determinant of physician performance, yet it plays a critical role in maintaining cognitive function, metabolic health, and circadian stability, all of which are challenged by shift work. Irregular schedules and busy clinical environments frequently lead emergency physicians to skip meals or rely on highly processed foods, which can produce fluctuations in blood glucose that contribute to fatigue, irritability, and impaired concentration.
- Meals that combine complex carbohydrates, protein, and healthy fats help maintain more stable energy levels and sustained cognitive performance compared with refined carbohydrate–heavy foods. In practice, bringing prepared meals or nutrient-dense snacks to shift can help avoid reliance on convenience foods during busy periods.
- Meal timing also influences circadian rhythms. Maintaining relatively consistent eating patterns can support metabolic regulation, while large meals during night shifts may worsen fatigue and gastrointestinal discomfort; many clinicians tolerate smaller, lighter meals overnight more effectively.
- Hydration and caffeine use also affect performance. Even mild dehydration can impair attention and reaction time, while moderate caffeine early in a shift may improve alertness. Excessive or late caffeine intake, however, can interfere with post-shift sleep and contribute to cumulative fatigue.
- Simple strategies such as planning meals, prioritizing balanced nutrition, maintaining hydration, and using caffeine strategically can meaningfully support cognitive performance and long-term health throughout a career in emergency medicine.
Bottom Line – > Ultimately, maintaining personal physiologic health should be viewed not as optional self-care but as a professional responsibility essential for sustaining high-quality emergency care over the course of a long career.