Podcast: Play in new window | Download
вАЬThis learning material is sourced from Emergency Medicine Cases ¬†and has been published here with permission as per Creative Commons copyright.вАЭ¬†
Cardiac arrest care has always been about the fundamentalsвАФhigh-quality CPR, timely defibrillation, and effective ventilation. But as our guest experts¬†Dr. Sheldon Cheskes¬†and¬†Dr. Rob Simard¬†make clear in this EM Cases update as we reflect on the latest 2025 AHA guidelines, the fundamentals are evolving in ways that challenge some of our most ingrained habits and assumptions. In Part 1 of this series, we take a deep dive into the practical bedside application of CPR, defibrillation, and ventilationвАФmoving beyond вАЬcookbookвАЭ algorithms toward a more nuanced, performance-driven approach. From rethinking pad placement and shock strategy, to interpreting ETCOвВВ in context rather than chasing arbitrary numbers, to recognizing that even subtle leaning on the chest during compressions can undermine outcomesвАФthis episode is packed with pearls that demand we recalibrate how we run resuscitations. We explore why measuring CPR qualityвАФand feeding that information back in real timeвАФis no longer optional but central to care. We unpack concepts like compression-adjusted ventilation, the role of arterial lines during arrest, and feedback devices. And perhaps most provocatively, we challenge traditional dogma and the questions: Is the two-minute cycle too rigid? Should we be shocking earlier? Is head up CPR a viable technique?¬† and much moreвА¶
Podcast production, sound design & editing by Anton Helman; Voice editing by Erik Krosby
Written Summary and blog post by Anton Helman March, 2026
Cite this podcast as: Helman, A. Simard, R. Cheskes, S. Cardiac Arrest Update: Beyond the 2025 Guidelines Part 1: CPR, Defibrillation and Ventilation. Emergency Medicine Cases. March, 2026. https://emergencymedicinecases.com/cardiac-arrest-update-cpr-defibrillation-ventilation. Accessed March 27, 2026
Why this update on cardiac arrest management matters
The 2025 AHA ACLS Guidelines reaffirm what saves lives in cardiac arrest: rapid recognition, high-quality CPR, early defibrillation for shockable rhythms, timely vasopressor use, and coordinated postвАУcardiac arrest care. Those pillars are not newвАФand thatвАЩs the point. Survival gains in many systems have come less from novel drugs and more from better execution: faster emergency activation, higher bystander CPR rates, earlier AED deployment, and tighter choreography during resuscitation. What the guidelines cannot fully capture is the nuance required when the algorithm becomes sparse. In the ED, the most consequential decisions often occur precisely where evidence is uncertain and the flowchart stops giving directionвАФespecially in VF after multiple shocks, epinephrine, and amiodarone with no ROSC. At that inflection point, the clinician has to think physiologically: Are we generating coronary perfusion pressure? Are we preserving it with compression fraction? Are we undermining ourselves with hyperventilation? Is VF truly shock-refractory or simply recurrent? Is our defibrillation technique actually delivering current through myocardiumвАФor just delivering electricity into a high-impedance chest?
This episode focuses on those bedside inflection pointsвАФthe places where small adjustments in timing, technique, and interpretation can plausibly change outcomes.
Quick list of whatвАЩs new since our¬†last episode on cardiac arrest
Chain of Survival
- The chain of survival now explicitly adds recovery and survivorship as a 6th link, emphasizing ongoing support after hospital discharge rather than ending care at ROSC or discharge.
- Upstream system-level emphasis on dispatcher-assisted CPR, video-assisted CPR, crowdsourced responder/AED apps such as PulsePoint/FirstAED, and drone AED delivery to shorten time to first shock.
- A stronger endorsement of stay-and-play over load-and-go for most out-of-hospital arrests, based on the idea that paramedics can do nearly everything on scene that the ED can do, and that arrival to the ED without ROSC carries a very poor prognosis.
- A systems concept of transporting postarrest patients to cardiac arrest centers / organized centers of excellence with regular exposure to arrest care, PCI capability, targeted temperature management, and specialized ICU/cardiology/neurology support.
CPR
- A stronger practical message that measuring CPR quality and feeding it back to teams is itself central; when CPR quality was first objectively measured, essentially no system was delivering truly high-quality CPR.
- Discussion of noninvasive flow assessment during arrest, including a carotid flow patch concept, with the caution that some devices measure velocity rather than flow, and those are not equivalent.
- More detailed bedside teaching on recoil of the chest: the code leader or CPR coach should visually watch for subtle leaning on the chest as compressors fatigue rather than relying only on rate/depth.
- Preference that the best CPR feedback devices are integrated into the defibrillator rather than standalone devices, because they are easier to use and can display rate, depth, rhythm, and now ventilation parameters in one place.
- The head-up CPR discussion is more skeptical: it emphasizes that head-up CPR is really a bundle of elevation + Impedence Threshold Device (ITD) + mechanical CPR, that the animal data are much stronger than the human data, and that Lucas migration on an incline is a major practical problem.
- The mechanical CPR should not be used routinely if you can already provide high-quality manual CPR, even during short transports.
- Mechanical CPR has a major learning curve and should ideally be applied within about 10 seconds, with вАЬpit-stopвАЭ style team training.
- Use mechanical CPR as an academic/training tool during selected resuscitations so teams can practice rapid deployment for the situations where it may truly matter.
Monitoring, resuscitation targets and ventilation
- A more nuanced approach to ETCOвВВ: do not chase a single number; interpret it in context, and specifically be cautious that narrow-complex PEA with a high ETCOвВВ may represent pseudo-PEA rather than true pulselessness. COвВВ retainers, bag-mask leaks / lack of a closed system, and uncertain prolonged downtime can all distort the value.
- If present, an arterial line is considered the gold standard physiologic monitor; one target is diastolic pressure >30 suggesting decent CPR, while sudden increases may suggest ROSC and very low values suggest poor prognosis.
- Some systems, especially in Europe, may target MAP during arrest rather than only diastolic pressure.
- Ventilation feedback devices are emerging and may be the next major frontier, displaying both ventilatory rate and tidal volume.
- Question the evidence basis for the traditional 500вАУ600 mL tidal volume target; active investigation into whether 300вАУ400 mL may be preferable.
- A practical ventilation pearl is compression-adjusted ventilation (CAV): using about 10вАУ12 compressions per breath to maintain one breath every six seconds.
- Discussion of early use of a ventilator in CPR mode to control asynchronous ventilation during arrest rather than relying on a manual bagger.
Defibrillation
- A common defibrillation mistake is bad pad position, especially placing the lateral pad too anterior and too inferior, effectively вАЬdefibrillating the spleen.вАЭ
- A major new concept is that for VF, current matters more than energy, and Antero-Posterior (AP) pad position may be physiologically superior to Antero-Lateral (AL) because it delivers more current through the ventricles.
- Distinguish Atrial Fibrillation (AF) cardioversion from VF defibrillation: AL has shown to be more effective than AL in AF because of the relevant atrial myocardium, whereas AP may be better for VF because recurrent VF often arises from more posterior-inferior ventricular myocardium.
- Practical logistics for AP defibrillation: teams can place AP pads with a quick log roll during ongoing resuscitation and some resuscitations now start AP first.
- After the first few failed shocks, POCUS can be used to check whether the heart is actually sandwiched between the pads, not for the first shock, but to optimize subsequent shocks.
- A challenge to the fixed two-minute cycle: most refibrillation happens within about 30 seconds, not two minutes, and argue that earlier re-shocking when refibrillation is seen may be more physiologic.
- Related to that, see-through CPR algorithms can filter out compression artifact and allow earlier recognition of refibrillation during ongoing CPR.
- The shock-energy discussion is updated toward a high-energy/high-current upfront strategy, e.g. repeatedly using the higher energy settings available on a given defibrillator rather than escalating slowly from lower energies.
- For hirsute patients, the practical вАЬwax jobвАЭ trick: apply pads, rip them off to remove hair, then place a fresh set or manual pressure augmentation over the pads to reduce transthoracic impedance.
Refractory and recurrent VF, Dual Sequence Defibrillation
- For refractory VF, a much more granular taxonomy: pragmatic refractory VF, true shock-refractory VF, and recurrent VF, and emphasize that recurrent VF should not automatically be assumed to be purely a drug/LAD problem rather than a defibrillation problem.
- The Dual Sequence Defibrillation (DSED) rationale is updated: it likely works not because it is simply вАЬmore juice,вАЭ but because it provides more homogeneous current distribution across the ventricle and because the second shock sees a different myocardial state/rhythm milliseconds after the first shock.
- In trained systems, DSED did not worsen CPR quality, with chest compression fraction remaining similar across DSED, vector change, and standard groups.
- DSED and vector change are not equivalent interventions, and if two defibrillators are available, the clear preference is DSED over vector change.
- Early DSED discussion: emerging European work suggesting a possible signal that earlier DSED, even after the first failed shock, may be beneficial, contrary to the idea that early DSED may be harmful.
- Stellate Ganglion block is a fairly simple procedure using PoCUS similar to placing a central line.
Epinephrine
- Our experts suggest minimizing epinephrine is VF arrests, and stopping epinephrine in refractory VF.
- There has been a de-emphasis on use of IO in cardiac arrest; in patients without IV access IM epinephrine is recommended by our experts over IO, unless rapid PoCUS confirmation of IO placement is feasible.
The Big Picture: What actually improves outcomes
Cardiac arrest care is defined less by what we add and more by how well we execute what already matters. The latest guideline-informed perspective reinforces that survival is driven by a series of interdependent steps, each of which must be optimized.
The chain of survival now explicitly includes:
- Early recognition and activation¬†вАУ Dispatcher recognition of arrest has improved, some systems have adopted video-assisted dispatcher coaching, citizen-responder smartphone activation has increased early CPR/AED retrieval, and AED registries integrated into dispatch platforms can shorten time to first shock. Emerging real-world pilots using drone-delivered AEDs suggest meaningful time savings in VFвАФbecause in VF, seconds are physiology.
- High-quality CPR
- Early defibrillation
- Advanced resuscitation
- Post-arrest care
- Recovery and survivorship
The greatest impact comes from the earliest linksвАФrapid activation, bystander CPR, and early defibrillation. By the time a patient reaches the ED without ROSC, prognosis is already significantly diminished, underscoring that ED care is only one part of a larger system.
High-quality CPR: Not so simple
High-quality CPR is one of the only interventions with a clear and consistent relationship to survival and neurological outcome. Even experienced clinicians frequently perform CPR poorly. CPR is physically demanding, and degradation in performance happens rapidly. Even within 45 seconds, compression quality begins to decline, which is why switching compressors every two minutes is not just recommendedвАФit is essential. What is often under-appreciated is that CPR is not simply about generating movementвАФit is about generating forward blood flow. Every componentвАФrate, depth, recoilвАФdirectly influences cardiac output during arrest. If any of these are suboptimal, perfusion to the heart and brain falls.
In practice, this means that the team leader must actively monitor CPR as a primary intervention, not assume it is being done correctly. High-performing teams treat CPR like a continuously titrated therapy. We often teach вАЬrate 100вАУ120, depth 5вАУ6 cm, full recoil,вАЭ but those are surrogates. The real goal is uninterrupted perfusion.
Which CPR targets have the strongest causal plausibility?
Aortic diastolic pressure/arterial lines #1
If you rank physiologic targets by direct mechanistic link to ROSC and defibrillation success, aortic diastolic blood pressure is the most causally plausible bedside surrogate for CPPвАФwhen you have an arterial line. Diastolic pressure directly reflects the pressure gradient driving coronary blood flow, and both animal and human physiologic data support the association between higher diastolic pressures during CPR and higher ROSC likelihood. In practical terms, if an arterial line is present, many experienced resuscitationists aim for¬†diastolic BP вЙ•25вАУ30 mmHg¬†(often higher if achievable) for whether CPR is generating meaningful myocardial perfusion.
The operational nuance for placing an arterial line is staffing and opportunity cost. In a lean resus (one physician and one nurse), arterial line placement can be counterproductive if it distracts from compressions, defibrillation timing, and ventilation discipline. With adequate personnel, an a-line can serve three high-yield roles:
- Objective CPR quality feedback
- Differentiation of pseudo-PEA/low-flow states from true no-flow
- A rational framework for vasopressor strategy (treat a measurable perfusion deficit rather than giving blind doses)
Pitfall:¬†Treating an arterial line like a life-saving intervention in itself. The line is a monitorвАФnot a therapyвАФand it must never cost compression quality or pause time.
End-Tidal COвВВ monitoring in cardiac arrest
ETCO2 has strong physiologic plausibility because it reflects pulmonary blood flow and thus cardiac output generated by compressions. Higher ETCOвВВ generally correlates with better perfusion and higher probability of ROSC, and a sudden sustained rise can signal ROSC. However, ETCOвВВ is indirect and confounded by ventilation, airway problems, lung pathology, and metabolic state. It is best used as a trend and a quality monitor rather than a rigid interventional endpoint.
Compression fraction¬†is foundational because it preserves CPP. It is not a вАЬphysiologic metricвАЭ in the same way, but it is causally upstream: if compression fraction is low, neither diastolic pressure nor ETCOвВВ can be trusted to improve meaningfully.
Pearl:¬†If you can measure only one physiologic CPR target, diastolic pressure has the strongest causal plausibility as a surrogate for CPP. ETCOвВВ is extremely useful, but it is an indirect surrogate and must be interpreted in context.
End-tidal COвВВ confounders:
- Airway leaks (BVM vs intubation)
- Baseline COвВВ retention
- Duration of arrest
- Quality of CPR
For example, a patient with chronically elevated COвВВ may have misleadingly high values, while a prolonged downtime may produce low values despite adequate CPR. The key clinical application is to interpret ETCOвВВ in context, rather than using it as a standalone decision-making tool.
Which ETCO2 numbers matter (and what they mean) in cardiac arrest
- ETCOвВВ persistently <10 mmHg after ~20 minutes¬†of high-quality CPR is associated with a very low likelihood of ROSC (particularly when reversible causes have been addressed).
- ETCOвВВ вЙ•20 mmHg¬†suggests more effective perfusion and is generally reassuring that compressions are generating meaningful forward flow.
- A¬†sudden sustained rise of вЙ•10 mmHgвАФoften into the¬†35вАУ45 mmHg rangeвАФshould prompt an immediate ROSC assessment (because this pattern frequently reflects return of perfusion).
When low ETCOвВВ should trigger action in cardiac arrest
A persistently low ETCOвВВ should trigger one question first: Is this a CPR quality problem, or a physiology problem?
If ETCOвВВ is <10вАУ15 mmHg:
- Re-optimize compressions immediately (depth, recoil, rate, minimize pauses, avoid leaning on the chest).
- Reassess ventilation rate and volume (hyperventilation is common and harmful).
- Consider reversible causes that reduce forward flow (massive PE, tamponade, profound hypovolemia) or severely deranged metabolic state.
Confounders: What falsely lowers or raises ETCOвВВ in cardiac arrest
Falsely low ETCOвВВ can result from hyperventilation, excessive tidal volumes increasing intrathoracic pressure, poor compressions, severe PE, low metabolic COвВВ production in prolonged arrest, airway leaks/disconnections, or severe obstructive lung disease. Falsely elevated ETCOвВВ can occur with hypoventilation, bicarbonate administration (transient COвВВ load), ROSC, or certain V/Q mismatch patterns.
Pitfall:¬†ETCOвВВ used as a discreet target is a common pitfall. Rather ETCO2 is a trend monitor. Use it to detect deterioration in perfusion, guide CPR optimization, and identify ROSC.
The most common CPR failures in cardiac arrest
Key recurring problems include:
- Compression rate outside the 100вАУ120/min target
- Inadequate depth
- Leaning on the chest, preventing recoil
- Failure to rotate compressors
- Hyperventilation
Each of these has a direct physiologic consequence. For example, leaning prevents ventricular filling during recoil, reducing preload. Incorrect rate disrupts optimal coronary perfusion pressure. Hyperventilation increases intrathoracic pressure, impairing venous return.
CPR errors are cumulative. Multiple small deviations can combine to produce profoundly ineffective circulation, even when CPR appears вАЬacceptableвАЭ at a glance.
CPR quality: Measurement drives performance in cardiac arrest management
When CPR quality was first objectively measured in large studies, no system was consistently delivering high-quality CPR. Only after introducing measurement and feedback did performance improve.
In practical terms, this has several implications:
- Real-time feedback devices improve adherence to targets
- Post-resuscitation debriefing identifies performance gaps
- Teams become more attentive to CPR as a monitored intervention
Minimizing CPR interruptions: Planning, not speed
Interruptions in CPR are physiologically devastating. Coronary and cerebral perfusion pressures fall rapidly during pauses, and it takes multiple compressions to rebuild them. Minimizing interruptions is not about rushingвАФit is about anticipation.
Effective teams:
- Prepare for pulse checks in advance
- Position hands and probes before compressions stop
- Charge the defibrillator during compressions
- Perform rhythm and pulse checks simultaneously
- Resume CPR immediately after shock
PoCUS-assisted pulse checks: what they add and what they risk
Ultrasound pulse checks can improve accuracy if performed with strict discipline. The value is not вАЬmore dataвАЭ; it is faster confirmation of pulsatility at a known anatomic target. The danger is that ultrasound becomes a pause-extending distraction. The probe must be ready, positioned during compressions, and used to confirm flow during a brief pauseвАФthen compressions resume immediately. If interpretation requires longer thinking, capture/record a short clip and interpret after compressions restart.
Pearl: To minimize CPR interruption record the PoCUS pulse check for 3-5 seconds and resume CPR immediately, then interpret the recording
CPR feedback devices
Feedback devices are appealing because they target something we know mattersвАФcompression qualityвАФbut the evidence is mixed and stronger for CPR process metrics than for survival. Real-time audiovisual feedback can modestly improve compression rate, depth, recoil, and sometimes compression fraction. Metronomes can help prevent drift in rate but do not ensure depth or recoil and have not convincingly improved outcomes alone. Analogue tactile вАЬclickerвАЭ devices show promising signals in low-certainty data, likely because they enforce adequate depth with an unmistakable cue.
In practice, the most defensible role for feedback devices is within a quality improvement program. Measuring CPR quality across arrests, reviewing performance, identifying recurring failure modes, and retraining accordingly is where devices may produce system-level benefit. Even sensor-only measurement can improve performance through observation effectsвАФteams compress better when they know compressions are being measured.
Head-up CPR: Makes sense, but is it pragmatic?
Head-up CPR has a physiologic rationale that is easy to like: elevating head and thorax may reduce intracranial pressure and improve cerebral venous drainage, potentially improving cerebral perfusion. But the concept is usually bundled with strategies to augment forward flow (active compressionвАУdecompression CPR, impedance threshold devices). Without those adjuncts, tilting could reduce venous return and compromise cardiac output.
Operationally, head-up CPR is difficult to do well with manual compressions. Maintaining consistent depth, recoil, and compression fraction while the patient is tilted is challenging. For practical reasons, head-up CPR typically needs to be paired with mechanical CPR to provide consistent compressions while allowing controlled elevation. The evidence base remains early and limited, with feasibility studies and small datasetsвАФoften bundled interventionsвАФmaking it hard to isolate which component drives any observed signal. The strongest evidence for head-up CPR comes from animal studies, where combining head elevation with an impedance threshold device and mechanical CPR shows clear improvements in cerebral perfusion and intracranial pressure. However, they are explicit that translating these results into humans has been problematic, and that human studies are small and conflicting, with no convincing demonstration of improved survival. Although one center (Minnesota) has reported success with head-up CPR, other systems have not been able to replicate those results,¬†raising concern that outcomes may depend heavily on highly specialized protocols and early implementation that are difficult to reproduce.
Mechanical CPR is recommended for specific situations
Mechanical CPR is not recommended for routine use, but it has clear roles in specific situations:
- Prolonged transport
- Limited personnel
- Difficult environments
A key limitation is that application of the device can cause significant interruptions if not performed efficiently. There is a clear learning curve, and teams must train to apply devices rapidly (ideally within 10 seconds).
Ventilation: The quiet underminer
Ventilation mismanagement is one of the most common iatrogenic harms during cardiac arrest. Hyperventilation increases intrathoracic pressure, reduces venous return, decreases CPP, and increases intracranial pressureвАФreducing cerebral perfusion. Even experienced providers hyperventilate during the adrenaline of a code. The recommended approach remains simple but hard to execute: once an advanced airway is in place, provide¬†one breath every 6 seconds (10/min), deliver each breath over ~1 second, and use only the tidal volume needed for visible chest rise. Over-ventilation is often not recognized in real time, which is why ventilation feedback devices and structured strategies are an important future direction.
Compression-Adjusted Ventilation (CAV): a practical solution
Instead of timing breaths by counting seconds (which often fails under stress), synchronize ventilation with compressions using a 12:1 compression-to-ventilation ratio (one breath after every 12 compressions). At a compression rate near 120/min, this naturally generates ~10 breaths/min and reduces drift into hyperventilation. It anchors behavior to the rhythm of CPR rather than the wall clock.
Where research is headed in ventilation strategy during cardiac arrest
Ventilation strategy during arrest is a major area of future research. The OPTIVO trial (Optimized Ventilation in Cardiac Arrest) is designed to evaluate whether a more physiologically optimized ventilation strategyвАФcontrolling rate and volume to minimize intrathoracic pressure and preserve venous returnвАФimproves outcomes compared with conventional practice. In parallel, ventilation feedback devices that provide real-time rate and tidal volume cues may reduce hyperventilation during chaotic resuscitation. Additionally, the evidence for the traditional 500вАУ600 mL tidal volume target is weak; there are active investigations into whether 300вАУ400 mL may be preferable. Early use of a ventilator in CPR mode to control asynchronous ventilation during arrest has a strong physiologic rationale but requires further study.
Pearl:¬†If you canвАЩt reliably deliver one breath every 6 seconds, use compression-adjusted ventilation (12:1) to вАЬhard-wireвАЭ the correct rate into the resuscitation rhythm.
Defibrillation strategy in cardiac arrest
Defibrillation is deceptively simpleвАФpush a button and shockвАФbut success depends on whether current actually traverses myocardium and whether perfusion has primed the heart to respond. Many вАЬby the bookвАЭ failures are not failures of the algorithm, but failures of technique and choreography.
The most common defibrillation mistakes
Peri-shock pause is the major culprit: stopping compressions too early, charging after the pause begins, delaying compressions after the shock, and allowing rhythm checks to sprawl into extended hands-off time. Technical errors also matter: poor pad adhesion, pads placed on hair or moisture, incomplete chest exposure, or pad placement that does not maximize current through ventricular myocardium. A final strategic mistake is repeating identical shocks indefinitely without escalating defibrillation conditionsвАФvector change, pad repositioning, energy escalation, or advanced strategies in refractory VF.
Shock timing and the вАЬtwo-minute cycleвАЭ
The two-minute CPR cycle is a practical teaching tool, not sacred physiology. Recurrent VF often occurs early after a shock within 30 seconds. Where rhythm filtering allows detection of refibrillation during compressions, there is physiologic plausibility to re-shock promptly when VF is clearly present rather than waiting an arbitrary full cycle. This is not stacked shocks with long pausesвАФit is the idea that we should avoid unnecessary time in VF while preserving compression fraction.
AP pad placement is preferred over AL
Emerging evidence suggests that anteriorвАУposterior (AP) pad placement may deliver more current through the ventricles compared to anteriorвАУlateral (AL). This is particularly relevant in ventricular fibrillation, where the goal is to deliver current through the ventricular myocardium. In contrast, atrial fibrillation cardioversion targets the atrium, explaining why AL may be ore effective for termination of atrial fibrillation.
Practically, AP placement can be achieved with minimal interruption:
- Continue compressions
- Briefly log roll to place posterior pad
- Place anterior pad
- Resume CPR and shock
Defibrillation: ItвАЩs about current, not just energy
A key conceptual shift is understanding that currentвАФnot energyвАФis what matters in defibrillation. This brings pad placement into focus. Incorrect placement, particularly placing the lateral pad too low or anterior (effectively shocking the spleen!), can result in ineffective current delivery. Many pad placements effectively вАЬmiss the heart,вАЭ delivering current through non-cardiac structures.
Pearl:¬†Start with the fastest, cleanest, most consistent pad placement that preserves compression fraction. Escalate to vector change deliberately when shocks failвАФwithout paying for it in pause time. Some systems start with AP positioning which appears to maximize current through the heart when compared to AL pad positioning.
Defibrillating hirsute patients (practical solutions)
In hairy patients, poor pad adhesion increases impedance and reduces effective current delivery. The key is to solve the contact problem without delaying shocks.
- Use the pads as a вАЬwax stripвАЭ: apply firmly, rip off quickly to remove hair, then place fresh pads
- Manually press down on the pads firmly to maximize skin contact during shock delivery (this has shown to be safe for the provider)
- Shave quickly only if adhesion remains impossible (avoid prolonged delay)
- Ensure chest is exposed and dry (avoid clothing/moisture barriers)
Pearl: Manually applying pressure to the pads in hirsute and/or sweaty patients during defibrillation is safe for the provider and increases current, the key factor in effective defibrillation
Refractory VF management
The 2025 AHA Guidelines provide no recommendations after 3 shocks, epinephrine and amiodarone are given in VF.
Timing of defibrillation: Challenging the 2-minute cycle in VF
The traditional two-minute CPR cycle is increasingly questioned. Most patients who refibrillate do so within 30 seconds, not two minutes. This raises concern that waiting a full cycle before re-shocking may delay effective therapy.
While evidence is not definitive, the physiologic argument supports:
- Earlier recognition of refibrillation
- Earlier re-shocking when appropriate
Dual sequential external defibrillation (DSED) rationale and current evidence
Dual sequential external defibrillation (DSED) uses two defibrillators and two pad sets to deliver two shocks in rapid sequence. This not simply вАЬmore joules,вАЭ but improved myocardial coverage through multiple vectors and a potential priming effect where the first shock alters the VF substrate, increasing susceptibility to the second shock delivered milliseconds later.
A major controversy is whether DSED harms CPR quality by introducing complexity and longer pauses. In the DOSE-VF trial, chest compression fraction was similar across all groups, including DSED. This suggests that, in trained systems, DSED can be implemented without compromising CPR quality, which is critical since CPR quality is strongly tied to outcomes. The practical implication is that DSED is a training problem, not an unavoidable pause problem: if your system cannot do it without prolonged hands-off time, it may not be worth it; if it can be integrated cleanly, it becomes a preferred escalation strategy.
The clinical trial evidence (DOSE-VF)
DOSE-VF trial is a high quality sizeable randomized trial comparing:
- Standard defibrillation
- Vector change (switching pad position from anteriorвАУlateral to anteriorвАУposterior)
- Dual sequential external defibrillation (DSED)
The trial demonstrated that both vector change and DSED improved outcomes compared to standard defibrillation, including higher rates of ROSC and improved survival. While both vector change and DSED performed better than standard care, DSED is favored when feasible according to our experts.
Timing of DSED: Earlier may be better
Traditionally, DSED has been reserved for refractory VF after three failed shocks. However, there exists emerging data suggesting that earlier use of DSED may be beneficial. There are several small European studies showing a signal that early DSEDвАФeven after the first failed shockвАФmight improve outcomes, and importantly, that early use does not appear harmful. Many institutions in North America already use DSED after the second shock, while others are waiting for more data before moving it as early as after the initial shock. This challenges the traditional вАЬlate rescue therapyвАЭ mindset and suggests that DSED may be more effective when used¬†before¬†prolonged ischemia and myocardial deterioration occur.
Stellate Ganglion Block in refractory VF
Rationale
Stellate ganglion block is grounded in a simple physiologic idea: sympathetic overdrive fuels electrical instability. In prolonged refractory VF or electrical storm physiology, the myocardium is often bathed in catecholaminesвАФfrom endogenous stress response and from exogenous epinephrine. This sympathetic surge lowers arrhythmia threshold and can perpetuate recurrent or refractory VF despite shocks and antiarrhythmics.
The stellate ganglion provides major sympathetic input to the heart. Blocking it produces a rapid temporary chemical sympathectomy, aiming to reduce arrhythmogenic drive and stabilize the myocardium so defibrillation and perfusion strategies can succeed. Observational evidence and systematic reviews suggest high rates of arrhythmia suppression or reduction in electrical storm, with low rates of serious complications. It is best thought of as a bridge therapyвАФbuying time for definitive management such as PCI, ECMO, or ablation.
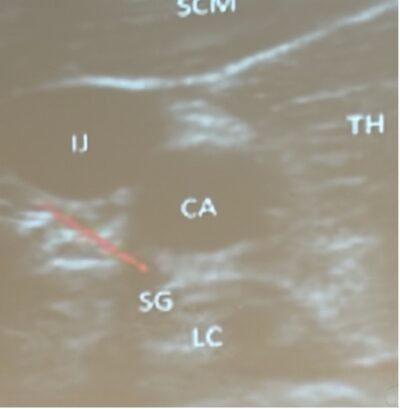
Stellate Ganglion Block Procedure
- Indication: refractory VF / suspected electrical storm physiology after optimized defibrillation strategy and standard meds
- Do not stop CPR: perform during ongoing chest compressions whenever feasable
- Use ultrasound guidance
- Identify left carotid artery
- Target location:¬†just inferior to the left carotid at roughly the 6 oвАЩclock position, typically around C6
- Injectate: ~20 mL lidocaine or bupivacaine into the appropriate fascial plane adjacent to the stellate ganglion under ultrasound visualization
- Goal: reduce sympathetic tone and ventricular irritability; suppress recurrent VF
Risks and considerations of stellate ganglion block
Complications are uncommon. Transient HornerвАЩs syndrome and hoarseness may occur. Serious complications (vascular injury, pneumothorax, major bleeding) are rare with ultrasound guidance. Evidence is largely observational and protocols vary, but the physiologic rationale is coherent and the bedside feasibility is high in trained hands.
Epinephrine nuances & vascular access in cardiac arrest
Epinephrine works because it raises aortic diastolic pressure via alpha-mediated vasoconstriction, increasing CPP and the probability of ROSC. The problem is that while epinephrine improves ROSC and survival to hospital admission, it has not consistently improved neurologically intact survival in a large, definitive way. That tradeoff matters most in shockable rhythms where defibrillation is the primary therapy and excessive catecholamine exposure may worsen electrical instability.
Dosing and the вАЬwhen to stopвАЭ conversation
Guideline dosing remains¬†1 mg IV/IO every 3вАУ5 minutes. High-dose epinephrine has not shown benefit and may cause harm. The nuanced bedside discussion is about rhythm-specific strategy: in VF, many clinicians stop epinephrine after 2вАУ3 doses if VF remains refractory or recurrent, especially when catecholamine-driven instability is suspected. In PEA/asystole, early epinephrine appears more clearly associated with improved ROSC, and continued dosing is reasonable while aggressively searching for reversible causesвАФthough prolonged futile resuscitations should still trigger a reassessment of goals and likelihood of meaningful outcome.
Pitfall: Continuing epinephrine for multiple doses without addressing reversible causes or without reconsidering whether catecholamines are perpetuating instability in refractory VF.
IM Epinephrine: A viable bridging strategy
IM epinephrine is being explored as a way to deliver earlier epinephrine when IV access is delayed, particularly in the prehospital setting. Observational and pilot data suggest shorter time to first dose and signals for improved ROSC and early survival, especially in non-shockable rhythms, with uncertain impact on neurologically intact survival. The physiologic concern is absorption: in a low-flow arrest state, skeletal muscle perfusion is reduced, making IM uptake potentially delayed or unpredictable. The practical stance is cautious optimism: IM may be a bridge when IV/IO is delayed, but it should not replace rapid vascular access until randomized outcome data clarify its effect.
IO Access: guideline direction, RCT reality, and PoCUS confirmation
The AHA guideline direction emphasized here is a practical hierarchy: IV preferred, IO as backup. The reason is not that IO never works, but that IO reliability and pharmacokinetics may be less consistent than previously thought, and RCTs have not shown superiority despite perceived faster placement. Malposition and silent failure are key concerns.
Where PoCUS adds real value is confirmation of IO placement. Doppler ultrasound during flush can confirm medullary flow quickly without interrupting CPR, identifying nonfunctional IOs that otherwise would be assumed to work.
Pearl:¬†If youвАЩre using IO in arrest, especially when expected drug effects are absent, PoCUS Doppler confirmation can convert IO from a hopeful access point to a verified delivery route.