פוסט זה זמין גם ב:
עברית
Podcast: Play in new window | Download
Emergency Medicine has undergone many changes over the last couple of decades and especially during the COVID pandemic. Most of these changes have been very positive, but increasing volumes, staff shortages, aging populations, increasing breadth of responsibilities and better access to more imaging have made some of us question how we should define the scope of our practices. In this main episode podcast, highlights from CAEP 2022 conference, Anton discusses the article ‘Saving EM: Is Less More?’ with Dr. Paul Atkinson and Dr. Grant Innes and offer some solutions to this current state of affairs in EM. In another CAEP highlight, trauma team leader Dr. Mathieu Toulouse delivers the latest on management of traumatic pneumothorax. We explore if less is more when it comes to indications for placing chest tubes, size of chest tubes, antibiotics etc. He answers such questions as: Do all patients with a traumatic pneumothorax require tube thoracostomy? How do CXR and CT differ in determining which patients require a chest tube? Do all patients receiving positive pressure ventilation require a chest tube for their traumatic pneumothorax? Does the presence of hemothorax necessitate placement of a chest tube? Are 14Fr pigtail catheters adequate for all traumatic pneumothoraces? and many more…
Podcast production, sound design & editing by Anton Helman
Written Summary and blog post by Hanna Jalali, edited by Anton Helman September, 2022
Cite this podcast as: Helman, A. Toulouse, M. Innes, G. Atkinson, P. Episode 174 Saving EM and Traumatic Pneumothorax – Highlights from CAEP 2022. Emergency Medicine Cases. September, 2022. https://emergencymedicinecases.com/saving-em-traumatic-pneumothorax. Accessed December 27, 2022
Saving Emergency Medicine—Is less more?
As EM physicians we pride ourselves in being able to provide care to any patient that steps through the ED doors. Historically, our care has adjusted to provide a wider breadth of knowledge, investigations, and procedures. However, considering the current state of ED including overcrowding with admitted patients, staff shortages and left-without-being-seen rates, it may be time to change our definition of what the scope of emergency department care is.
ED Overcrowding
ED overcrowding is one of the core current and projected concerns for being able to provide the excellent care that emergency physicians do. The problem is not necessarily with the ED itself but rather overall hospital crowding and access block. We currently seem to function under a model where nearly all unscheduled healthcare takes place in the ED.
- The Canadian Foundation of Health Improvement (CFHI) point out that ED visits are increasing at a much faster rate than population growth and are projected to grow an additional 40% by 2043
- There is dependency by Canadians on EDs with the highest rates of ED utilization in the developed world
- Canada performs worst among OECD countries in accessibility to primary care, specialists, surgical procedures, and imaging
Causes of ED Overcrowding: Access Block—Wrong Care, In the Wrong Place
Access block is defined as inability to access the care you need when it is needed. This can be considered as the “wrong care, in the wrong place, by the wrong provider, at the wrong time.”
We have come to accept that the emergency medicine consultant will be available for any unscheduled care, or simply when it is not convenient. This is not to say that EM physicians will be unavailable for unexpected emergencies – this is our core responsibility. There are predicted complications of care that are not emergent but are urgent or semi-urgent. There needs to be an alternative pathway for the clearly stable patient to get the “right care in the right place.”
There is general misallocation of care to the ED when patients have access block to primary care, imaging, or specialists.
Contributors to Access Block
- Patient complexity: EM training and ED visits are geared towards short interactions to stabilize and treat acute conditions; there is a concern for failure of ideal care in our medically complex and elderly patients for whom EDs are not well designed
- Staff shortages: Many staff including nurses and physicians are exiting the ED due to burnout; this has led to staff shortages that further increase the stress on our system and creates a vicious cycle
- Access to follow-up resources, primary and specialist care: in many jurisdictions the ED is the only place for a patient to go with a rising burden of unmanaged disease
- Shortfall of inpatient hospital space and staff
Potential Solutions to Access Block and ED Patient Boarding: Saving EM
A study of 25 Canadian hospitals showed on average that there are 46,000 hours of high acuity access block per site per year. The highest acuity patients are waiting to get a care space for a cumulative 46,000 hours per site per year. Admitted, stabilized, or diagnosed patients are gradually progressed to other levels of care in the hospital while those who are most at risk and could benefit the most from immediate investigation or intervention are too often left waiting in waiting rooms or ED hallways. We are missing 46,000 hours of space and staff per site which is a time where many adverse events can occur.
One solution is getting 46,000 hours of patient care space and staff in each hospital. Some suggestions for getting these extra hours include:
- Decreasing inpatient length of stay (LOS) by just 90 minutes, as the average hospital discharges 40,000 patients per year
- Reducing consult delay from ED to inpatient setting by 2 hours
- Increasing ED efficiency and reducing discharge LOS by 1 hour
Ultimately, what this study potentially shows is that much of what we see in ED overcrowding is a problem that can be solved without new capacity if there is onus placed on each non-ED service/program for efficiency and accountability to their patients. At each level, and transition between levels of care there are bottlenecks that the ED on its own cannot solve, but with collaboration between programs we can start to address. When the ED is overcrowded, EMS cannot provide the needed care to the community; when there is no access to inpatient hospital beds and patients are in stretchers the ED cannot provide adequate care to arriving patients; and so on. An accountability framework for flow must me developed at hospital administration levels with clear objective goals in place. Accountability for admitted patients that are not actually admitted is key. Currently, in patient floors/programs determine if they cannot manage their queue; the default is to stop the inflow of patients and close the front door. The resultant demand and failures in care are not seen and this halts innovation for efficiency. This breaks the accountability framework.
Summary of Solutions for ‘Saving EM’
- Reframe what emergency medicine is; redefine it’s scope of practice. There are emergencies waiting while the ED team deals with anticipated failures of our system. Our skillset needs to be recognized and redefined to protect emergent patients that are currently lost in the volume.
- Create and encourage others to respect an accountability framework. Encourage healthcare providers that are sending patients to the ED to not do so without speaking to us first. This will allow discussion for alternatives, or a plan prior to this patient facing a long wait.
- Funding, and incentives for other programs to see patients during non-banking hours and offload ED care.
Calls to Action for EM clinicians, leadership, and hospital administration
- Create accessible resources during non-banking hours
- Training: ED clinicians need skillset and space for primary care and dealing with complex and chronic disease patients; there is also a need for training within specialty and primary care programs that deals with unscheduled care
- ED leadership: we are the front lines and understand many of the healthcare gaps; we should involve yourselves and speak with hospital leadership, share our perspective, and advocate to implement positive change
- Compassion: for colleagues, patients, staff and for yourself. Reflect on your interactions and appreciate the level of care you can provide to our patients in need
Traumatic Pneumothorax Assessment and Management – Is Less More?
There is now very clear evidence that not all traumatic pneumothoraces require drainage. Tube thoracostomies are associated with complication rates of 10-30% and may cause significant pain to our patients which can impact rehabilitation.
Source: Western Trauma Association guidelines on management of pneumothorax
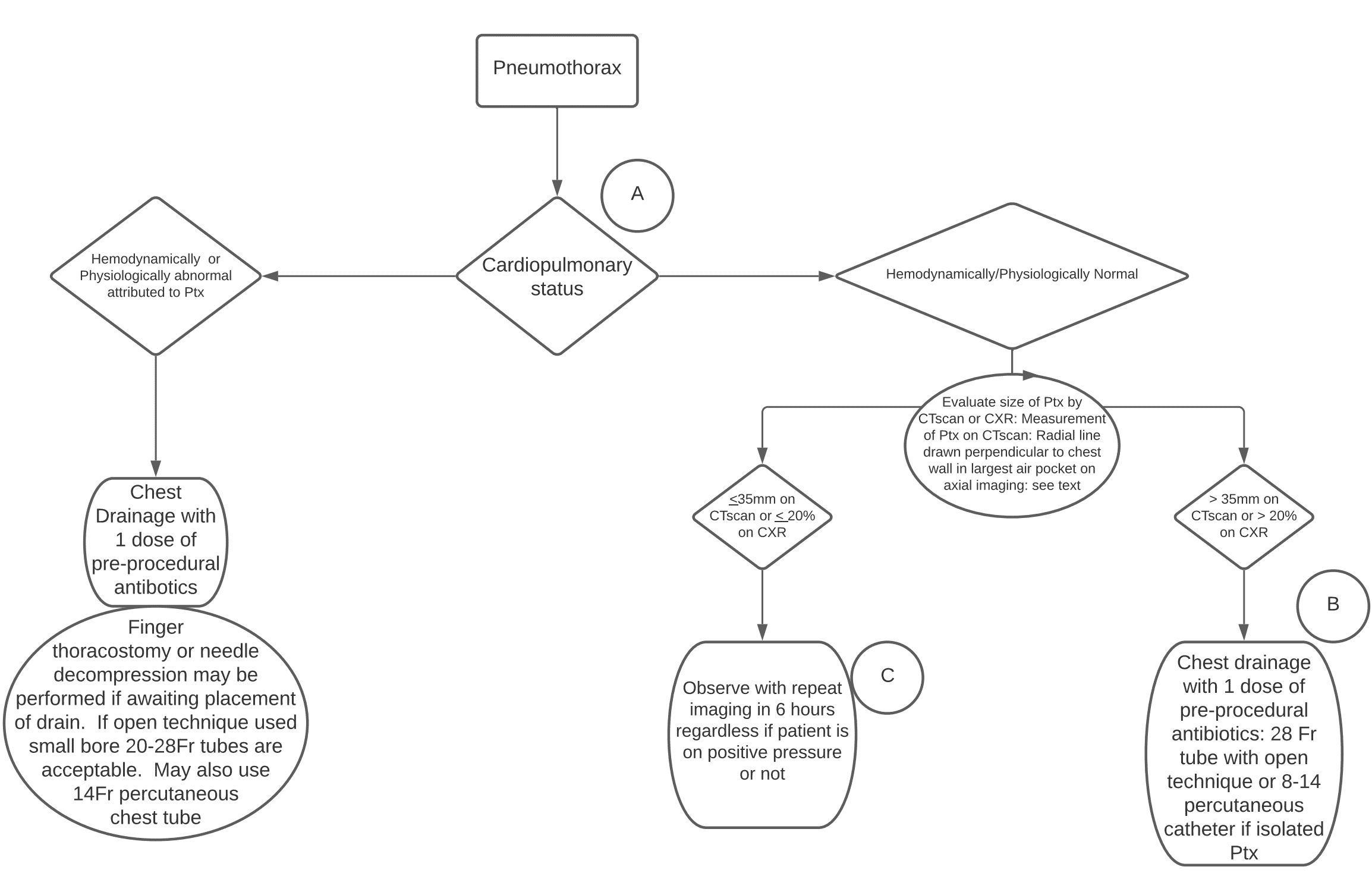
Which patients with a traumatic pneumothorax require tube thoracostomy?
The decision to place a chest tube is based on two questions:
- What is the cardiopulmonary status? Stable vs. unstable.
If the patient is hemodynamically or physiologically abnormal as a direct result of the pneumothorax, then a chest tube should be placed. If the patient is unstable and you are not sure whether it is due to the pneumothorax, the safe approach is to place a chest tube. In the crashing patient finger thoracostomy or needle decompression should be considered while waiting for placement of the chest tube.
- If stable, what is the pneumothorax size?
In general, the trauma literature suggests that smaller pneumothoraces have a better chance of observation success compared to larger ones.
Western Trauma Association guideline suggestions for indications to place a chest tube for traumatic pneumothorax in the stable patient
On chest x-ray: >20% pneumothorax
Reminder: Percentage calculations are not sensitive in supine patients and upright PA chest x-rays should be used to calculate the size. If you observe a 20% pneumothorax on supine x-ray, then it is likely much larger, and a chest tube should be placed.
On CT scan: >35mm
Measurement: A line in the largest air pocket on axial imaging drawn perpendicular or radial to the chest wall.

Measurement of size of pneumothorax on CT scan – line in the largest air pocket on axial imaging drawn perpendicular or radial to the chest wall; source: Western Trauma Association pneumothorax guidelines
≤ 35mm traumatic pneumothorax on CT predicts a success rate of 90% with observation; >35mm predicts a failure rate of 40%.
Considerations in patients with pneumothorax who require positive pressure ventilation (PPV)
The OPTICC trial looked at occult pneumothorax to chest x-ray in ventilated patients with a composite outcome of respiratory distress and concluded that those patients who require short term PPV (ie. for OR) have no significant difference in outcomes. Patients who require prolonged PPV for greater than 4 days (ie. ICU with PPV) have a 40% failure rate and chest tube placement is recommended for these patients.
The decision to place a chest tube in a patient on PVV with a traumatic pneumothorax should ultimately be made in concordance with your team and the accepting service, as close observation to detect early progression to tension pneumothorax is necessary for patients with pneumothorax on PPV who do not have a chest tube in place.
Factors to take into consideration in the decision to place a chest tube in ventilated patients with traumatic pneumothorax include:
- Prolonged transport times
- Situations where close observation for progression to tension pneumothorax is not possible
- Mechanism of injury – penetrating trauma patients who are ventilated tend to have higher rates of observation failure compared to non-ventilated patients in the setting of occult pneumothorax
Chest tube size for traumatic pneumothorax – bigger is not better
In the past, larger chest tubes (28-36Fr) were generally used for traumatic pneumothoraces due to concern of occlusion with blood. Recently, however there has been a shift to using smaller catheters including 14Fr pigtail catheters based on small RCTs and observational studies.
The average distance between ribs is 9mm; larger chest tubes (28-Fr and up) have diameters >9mm. This may cause the chest tube to compress the neurovascular bundle and cause significant pain to the patient.
Although there are no large RCTs, most evidence suggests that small conventional chest tubes (ie. 20-Fr) and even 14-Fr pigtail catheters are equally as effective for drainage of traumatic pneumothorax when compared to larger chest tubes. Many centers are now using pigtail catheters which tend to impart less pain and allow for quicker rehabilitation compared to larger chest tubes. The Eastern Association for the Surgery of Trauma (EAST) has a conditional recommendation in favour of using a pigtail catheter and a survey completed by them suggested that in stable patients the majority of surgeons would choose 14-Fr pigtail catheter and another 20% would use conventional 20-Fr chest tube.
Does the presence of hemothorax necessitate placement of a chest tube?
A 2021 multicenter prospective RCT totalling 119 patients comparing 14Fr Pigtail catheters to 28-32Fr chest tubes for management of traumatic hemothorax (excluded patients who were in extremis and required emergent tube placement) looking at the failure rate, defined as a retained hemothorax requiring a second intervention, found that failure rate, was similar between the two groups (11% pigtail catheters vs. 13% large chest tubes).
Bottom line: as per the WTA algorithm use the smallest bore chest tube available, even when hemothorax is present, unless the patient with a hemothorax/hemopneumothorax is unstable
Clinical pearl: The average distance between ribs is 9mm; larger chest tubes (28-Fr and up) have diameters >9mm. This may cause the chest tube to compress the neurovascular bundle and cause significant pain to the patient.
Peri-procedural antibiotic use for traumatic pneumothorax – should antibiotics be given routinely when placing a chest tube?
There is currently largely heterogenous evidence with no clear conclusion as to whether or not antibiotics are indicated for peri-procedure chest tube placement. The main complications that antibiotics are meant to prevent are empyema and pneumonia that may result after chest tube insertion.
- An RCT from 2014 comparing 24 hours of cefazolin vs placebo in blunt trauma showed no difference in complication rates
- EAST states that there is insufficient evidence, and this will continue to be the case until there is a large multi-center RCT available
- AAST observational study: no significant difference in complications or ICU LOS in those that received antibiotics or not
- WTA recommends one dose of pre-procedural antibiotics based on a meta-analysis that concluded a significant difference, but the papers used within the meta-analysis were heterogenous
- The most recent meta-analysis (2021) showed a significant difference in complication rates for penetrating trauma but not blunt trauma; in the context of a dirty open wound, antibiotics are likely to have more benefit.
Bottom line: Prioritize other lifesaving and proven outcome-changing interventions over administration of antibiotics, as currently no clear evidence for benefit exists. In the setting of penetrating trauma, consider that antibiotics may be more likely to be beneficial. Further evidence is required to determine the effect on antimicrobial stewardship and number needed to treat.
Take home points for traumatic pneumothorax assessment and management
- Unstable due to pneumothorax? Place chest tube for drainage of pneumothorax.
- Finger thoracostomy or needle decompression can be performed while waiting for chest tube placement.
- Consider 1 dose of pre-procedural antibiotics, especially in penetrating trauma, but this is low priority.
- Place the smallest bore chest tube available unless there is a significant hemothorax present
- Stable? Consider the size of the pneumothorax:
- If a radial line perpendicular to chest wall on axial imaging is ≤35mm on CT or ≤20% on upright CXR, observe with repeat imaging in 6 hours regardless of need for PPV. Otherwise, place a small chest tube, 20Fr or 14Fr Pigtail catheter, even if a hemothorax is present.